12-Mar-2021
Several vaccines have been developed in recent months to combat the ongoing COVID-19 pandemic, caused by SARS-CoV-2. Whilst these have been effective in neutralising the initial SARS-CoV-2 virus, many variants have appeared across the globe. This is as a result of high transmission rates and error-prone viral replication, leading to high rates of mutation.
Whilst many of these mutations have little to no effect on the fitness and transmission rate of the virus, some do – this has led to several highly transmissible SARS-CoV-2 variants, including the B.1.1.7 variant initially identified in Kent, UK.
Highly transmissible SARS-CoV-2 variants have often convergently evolved to share a common alteration to their receptor binding domain (RBD): the N501Y mutation. This is a change at the 501 residue of the ACE2 binding receptor from N to Y, which has been found to increase the binding of the virus to cells and is therefore likely to be the cause of its enhanced transmission rate.
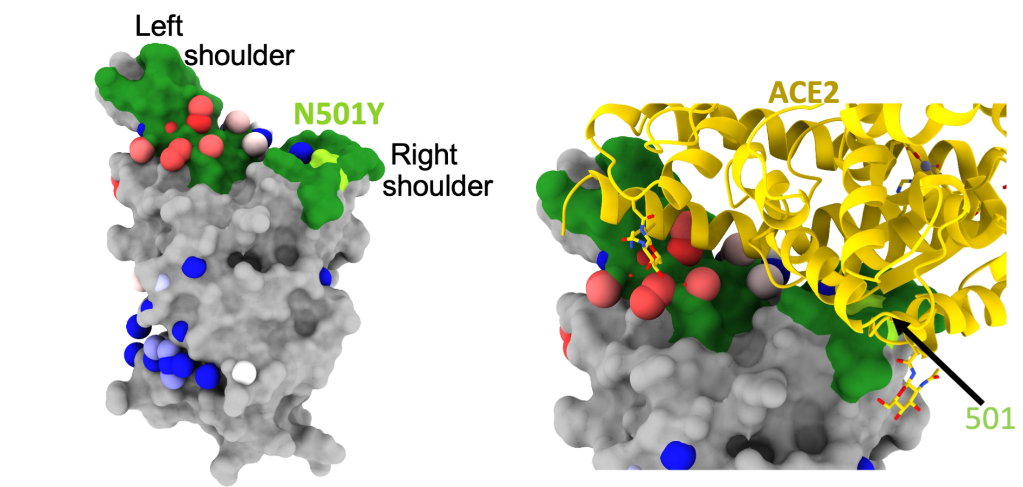
Figure 1. The 501 residue of the ACE2 binding receptor, located on the “Right Shoulder” of the protein, as analogised as a human torso. The 501 residue has changed from N to Y in variant strains of SARS-CoV-2.
This study investigated whether the B.1.1.7 variant was neutralised more or less effectively than the original viral strain using the most potent monoclonal antibodies identified by (Dejnirattisai et al., 2020), and by the currently rolled-out vaccines in the UK.
Utilising technology and laboratories from Instruct Centres UK, Diamond Light Source and STRUBI, the study determined that the majority of mAbs still bound the spike protein, unaffected by the N501Y mutation. However, some antibodies were dramatically affected, experiencing 78% and 100% reduction in binding in two cases.
In addition, neutralisation of the B.1.1.7 variant was tested using convalescent sera from patients immunised with current UK vaccines, produced by Oxford-AstraZeneca and by Pfizer. The convalescent plasma, taken from patients between 4-9 weeks from infection with the original strain, showed reduced neutralisation efficiency against the B.1.1.7 variant of 2.9-fold.
Similarly, vaccines raised against the original strain were less effective in protecting against the B.1.1.7 variant, although some viral neutralisation was still achieved. For convalescent plasma taken from patients infected with the B.1.1.7 variant, there was no significant difference in efficacy against the B.1.1.7 variant compared to the original strain.
In summary, this study found that the B.1.1.7 variant is more transmissible than the original strain, and that binding by antibodies (and hence neutralisation) in people infected with the original viral strain is affected. However, the vaccines currently in place in the UK, as well as antibodies built up by those who have recovered from the original strain, will neutralise the B.1.1.7 variant if present at slightly higher concentrations.
Read the full article: Supasa, P., Zhou, D., Dejnirattisai, W. et al. Reduced neutralization of SARS-CoV-2 B.1.1.7 variant by convalescent and vaccine sera. Cell (2021). DOI:https://doi.org/10.1016/j.cell.2021.02.033